Liquid Biopsy (LBx)
We employ blood-based liquid biopsy approaches to analyze the molecular characteristics of diseases and to develop precision diagnostics and companion diagnostics (CDx) technologies.
Conventional tissue-based diagnostics are limited in that they reflect tumor characteristics at a single time point and often fail to capture tumor heterogeneity and temporal changes in disease progression. In contrast, liquid biopsy enables the non-invasive and longitudinal monitoring of molecular alterations by analyzing diverse biomarkers present in the blood and is therefore emerging as a next-generation diagnostic approach.
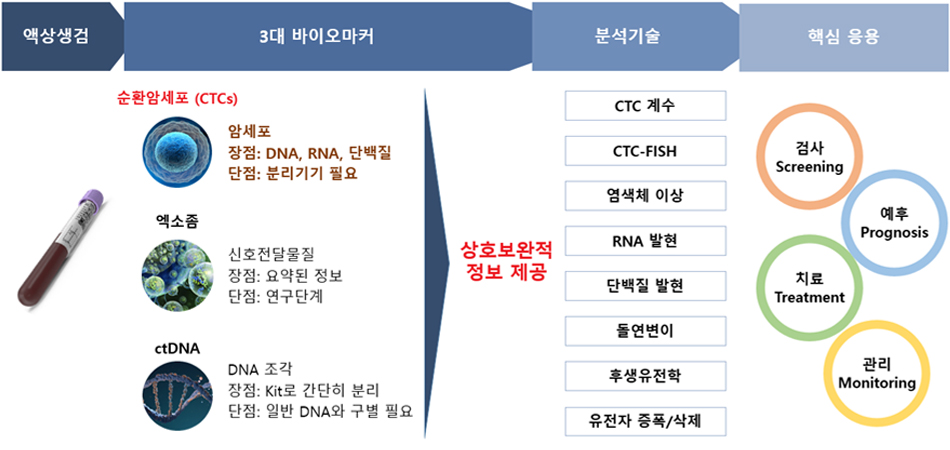
Accordingly, our research focuses on the integrated analysis of multiple liquid biopsy components—including circulating tumor cells (CTCs), plasma biomarkers, platelets, extracellular vesicles (exosomes) and immune cells—to elucidate disease-specific molecular features. These efforts aim to advance precision medicine and facilitate the development of companion diagnostics through liquid biopsy–based strategies.
1. Goal
Our research aims to establish a precision molecular diagnostic platform based on liquid biopsy technologies to elucidate the molecular characteristics of diseases.
This platform enables early disease detection, prediction of therapeutic response and the development of patient-specific treatment strategies.
Through blood-based non-invasive diagnostic approaches, we seek to introduce a new paradigm in precision medicine.
2. Research platform
We aim to establish an integrated liquid biopsy platform that enables the simultaneous acquisition and analysis of diverse molecular information from a single blood specimen.
Through the isolation and characterization of multiple liquid biopsy components, this platform facilitates a comprehensive understanding of disease-specific molecular alterations and the immune microenvironment.
2-1) Circulating tumor cell molecular profiling
Circulating tumor cells (CTCs) serve as important liquid biopsy biomarkers that capture tumor molecular characteristics and heterogeneity. Leveraging the GenoCTC platform, we isolate high-purity CTCs from peripheral blood and perform single-cell–level molecular profiling.
Specifically, we employ RNAscope-based in situ RNA analysis and cyclic immunofluorescence (CyCIF)–based multiplex protein analysis to comprehensively characterize gene and protein expression patterns.
These strategies facilitate in-depth elucidation of tumor heterogeneity and molecular features.
2-2) CTC-derived organoid platform
Due to the rarity of circulating tumor cells (CTCs), functional analyses are inherently limited. To address this challenge, we establish CTC-derived organoid (CDO) models by culturing CTCs under three-dimensional conditions.
These organoid models are utilized to assess anti-cancer drug responses, investigate mechanisms of drug resistance, and analyze tumor phenotypic plasticity. Ultimately, this research aims to establish a patient-specific therapeutic platform that integrates CTC isolation, organoid generation, and drug screening.
2-3) Platelet immune profiling
Platelets are key regulators of the tumor microenvironment and immune responses. We aim to identify novel liquid biopsy biomarkers associated with immunotherapy response by profiling platelet-associated immune markers.
Using flow cytometry, we analyze platelet activation markers, immune checkpoint–related molecules, and markers involved in immune interactions and co-stimulation. These approaches allow us to investigate the relationship between platelet characteristics and immune cell activation, as well as their impact on anti-tumor immunity.
2-4) Plasma biomarkers for neurodegenerative diseases
Our research focuses on the early detection and risk assessment of neurodegenerative diseases through the use of blood-based biomarkers. In particular, we analyze plasma biomarkers that reflect the pathological alterations associated with Alzheimer’s disease.
In addition, we leverage protein and nucleic acid information derived from plasma extracellular vesicles to enable more comprehensive and high-resolution characterization of the molecular features of neurodegenerative diseases.
2-5) Circulating fetal cell-based prenatal diagnostics
Our research focuses on developing next-generation non-invasive prenatal diagnostic technologies using circulating fetal cells (CFCs) derived from maternal blood. To address the limitations of conventional cfDNA-based assays, particularly maternal DNA contamination, we selectively isolate fetal cells and perform single-cell genomic analyses.
We aim to establish a next-generation non-invasive prenatal diagnostic platform by integrating microfluidics-based cell isolation, antibody-based cell selection strategies, and single-cell genomic profiling.
3. Clinical Research
Our research focuses on evaluating the clinical utility of liquid biopsy–based biomarkers through diverse clinical cohort studies by integrating blood-based molecular markers with patient clinical information. Through this approach, we aim to elucidate their associations with disease recurrence, therapeutic response, and clinical outcomes.
3-1) HER2-positive locally advanced breast cancer pCR study
Our research involves a prospective pilot study in patients with HER2-positive locally advanced breast cancer receiving neoadjuvant chemotherapy. Circulating tumor cells (CTCs) are longitudinally measured in blood samples obtained before and during treatment, and their association with pathologic complete response (pCR) is investigated.
By integrating CTC enumeration and phenotypic profiles with clinical data, we assess their predictive value for pCR. Ultimately, this study aims to validate the potential of CTCs as biomarkers for treatment response monitoring and clinical application.
3-2) Ongoing and planned liquid biopsy studies
Our research focuses on evaluating the clinical utility of liquid biopsy–based biomarkers across various cancer types through clinical studies. We are further expanding our efforts in predicting treatment response, identifying biomarkers associated with treatment-related toxicity and clinical deterioration, and analyzing tumor heterogeneity and therapeutic resistance.
Future studies will employ an integrated liquid biopsy approach that enables the simultaneous analysis of multiple liquid biopsy components from a single blood specimen, facilitating longitudinal monitoring of molecular alterations throughout cancer treatment.
