Cell and Gene Therapy (CGT)
1. CAR-X immunotherapy
CAR-X immunotherapy encompasses a range of therapeutic approaches in which immune cells are genetically engineered to express chimeric antigen receptors (CARs) and further optimized through advanced engineering strategies. These therapies are classified according to immune cell type, such as CAR-T and CAR-NK cells, as well as by their origin, including autologous and allogeneic approaches.
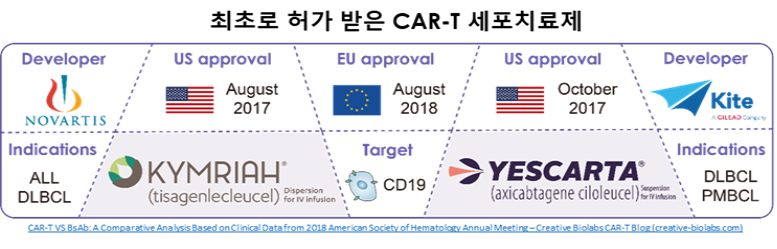
In 2017, Kymriah, the first CAR-T cell therapy, received regulatory approval following the demonstration of significant clinical efficacy in hematologic malignancies. Since then, CAR-based immunotherapies have rapidly advanced with extensive research efforts aimed at improving their efficacy and expanding their clinical applications.
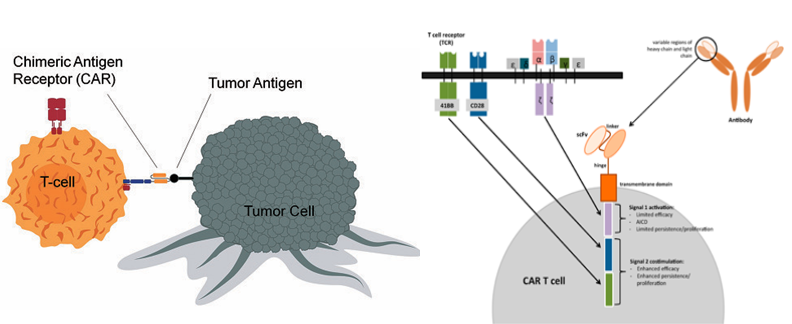
2. Chimeric antigen receptor ( CAR )
Chimeric antigen receptors (CARs) are synthetic receptors engineered to enable immune cells to recognize tumor-associated antigens. A typical CAR structure comprises an antigen-binding domain, a transmembrane domain, co-stimulatory domains that enhance activation and proliferation and an intracellular signaling domain that triggers immune effector functions.
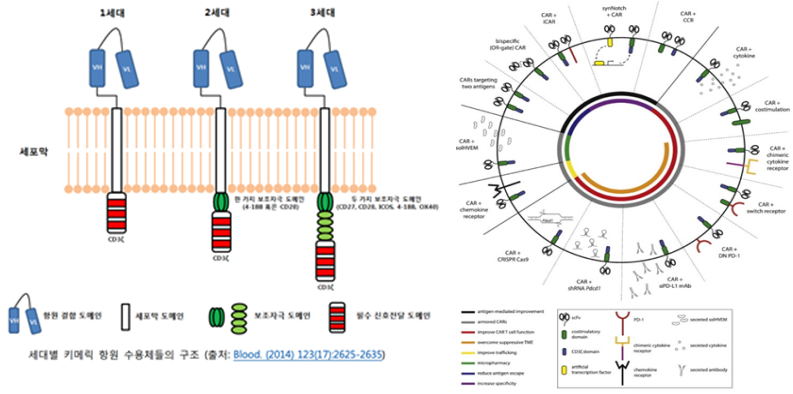
Advancements across CAR generations (first to third) have focused on the incorporation of additional co-stimulatory domains, thereby improving in vivo expansion, persistence and therapeutic efficacy. These modifications allow a limited number of infused cells to undergo robust proliferation and exert sustained anti-tumor effects.
In addition next-generation CAR designs are being developed to address existing limitations including dual-targeting CARs that recognize multiple antigens and inducible or conditionally active CARs that respond to defined environmental cues.
3.Trends in CAR immunotherapy
Approved cell-based therapies to date include CAR-T products such as Kymriah and Yescarta. CAR-X therapies have demonstrated substantial clinical efficacy in hematologic malignancies and are emerging as a promising next-generation therapeutic modality.
The global CAR-T market is anticipated to expand rapidly, growing from approximately USD 72 million in 2017 to an estimated USD 8.3 billion by 2028, corresponding to a compound annual growth rate (CAGR) of 53.9%. While CAR-T therapies are expected to dominate the immune cell therapy market in the near future, research and development efforts are rapidly advancing worldwide, including in Korea, where progress toward clinical application and commercialization is accelerating.
Furthermore, CAR-based therapies are being explored beyond oncology with expanding applications in autoimmune diseases and other indications.
4.Challenges in CAR-T cell therapy
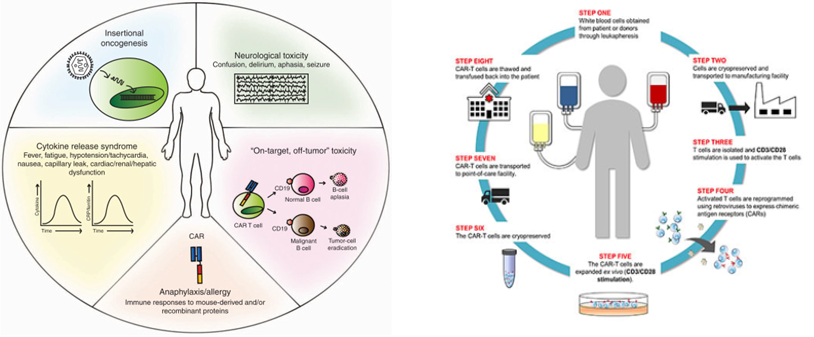
Approved CAR-T cell therapies to date rely on autologous T cells derived from individual patients requiring a complex and individualized manufacturing process involving cell isolation, genetic modification, expansion, and quality selection prior to reinfusion.
This process not only contributes to high treatment costs but also reflects the technical challenges associated with genetic engineering of primary immune cells, thereby limiting the integration of more advanced modifications.
While CAR-T therapies have shown substantial efficacy in hematologic malignancies, their application to solid tumors remains limited. Furthermore, critical challenges remain including severe toxicities such as cytokine release syndrome (CRS) and safety concerns related to viral vector–mediated gene delivery including the risk of insertional mutagenesis and oncogenesis.
These limitations underscore the ongoing need for innovative strategies to improve safety, broaden clinical applicability and enhance therapeutic outcomes.
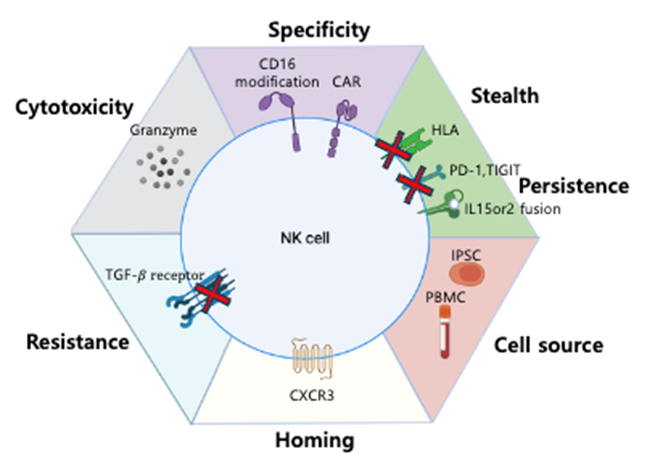
5. Current cell therapy research in our lab
Our research focuses on the development of next-generation cell therapies based on induced pluripotent stem cells (iPSCs). We have established robust differentiation and expansion protocols to generate diverse immune and regenerative cell types from iPSCs including natural killer (NK) cells, T cells, macrophages, microglia, and mesenchymal stem cells (MSCs) and are advancing their therapeutic applications.
iPSCs allow iterative genetic modification and selection enabling flexible genome engineering and functional optimization. These features provide significant advantages in scalability and versatility for cell therapy development.
By leveraging iPSC-based platforms, we aim to address key limitations of conventional autologous CAR-T therapies including complex manufacturing processes, high costs and variability in manufacturing success rates.
We are particularly focused on developing allogeneic, “off-the-shelf” cell therapy platforms in which cells derived from a single donor can be expanded, banked and used for multiple patients. This approach represents a transformative paradigm in cell therapy.
Our current efforts emphasize NK cell–based therapies including iPSC-derived NK cell differentiation and expansion strategies to enhance proliferative capacity along with additional functional engineering approaches as illustrated in the figure.
